





Updated on May 29, 2026
Photoplethysmography (PPG) technology has been used to develop small, wearable, pulse rate sensors. These devices, consisting of infrared light-emitting diodes (LEDs) and photodetectors, offer a simple, reliable, low-cost means of monitoring the pulse rate noninvasively. Recent advances in optical technology have facilitated the use of high-intensity green LEDs for PPG, increasing the adoption of this measurement technique.
In this review, we briefly present the history of PPG and recent developments in wearable pulse rate sensors with green LEDs. The application of wearable pulse rate monitors is discussed.
It is important to monitor the perfusion of the circulation. The most important cardiopulmonary parameter is blood pressure, but monitoring it is complicated. A second important parameter is blood flow, which is related to blood pressure. We can monitor the blood perfusion in large vessels using ultrasound devices, but it is not practical to use these routinely. Several devices for monitoring blood perfusion have been developed [1], but, unfortunately, it is difficult to find a practical device. However, the perfusion of blood flow and blood pressure can be determined easily using a pulse rate monitor. Wearable pulse rate sensors based on photoplethysmography (PPG) have become increasingly popular, with more than ten companies producing these sensors commercially. The principle behind PPG sensors is optical detection of blood volume changes in the microvascular bed of the tissue.
The sensor system consists of a light source and a detector, with red and infrared (IR) light-emitting diodes (LEDs) commonly used as the light source. The PPG sensor monitors changes in the light intensity via reflection from or transmission through the tissue. The changes in light intensity are associated with small variations in blood perfusion of the tissue and provide information on the cardiovascular system, in particular, the pulse rate. Due to the simplicity of this device, wearable PPG pulse rate sensors have been developed. This review describes the basic principles of PPG, previous and current developments in wearable pulse rate monitors with a light source, and the elimination of motion artifacts.
The principle of PPG has been reviewed previously [2–4], and is explained briefly here. Light travelling though biological tissue can be absorbed by different substances, including pigments in the skin, bone, and arterial and venous blood. Most changes in blood flow occur mainly in the arteries and arterioles (but not in the veins). For example, arteries contain more blood volume during the systolic phase of the cardiac cycle than during the diastolic phase. PPG sensors optically detect changes in the blood flow volume (i.e., changes in the detected light intensity) in the microvascular bed of tissue via reflection from or transmission through the tissue.
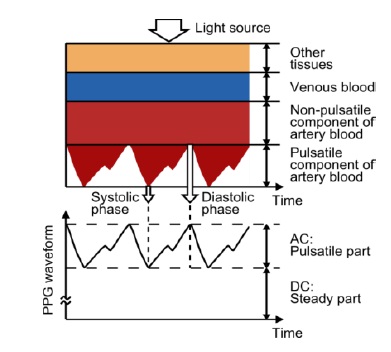
Figure 1 shows an example of a photoplethysmographic waveform, consisting of direct current (DC) and alternating current (AC) components. The DC component of the PPG waveform corresponds to the detected transmitted or reflected optical signal from the tissue, and depends on the structure of the tissue and the average blood volume of both arterial and venous blood. Note that the DC component changes slowly with respiration. The AC component shows changes in the blood volume that occurs between the systolic and diastolic phases of the cardiac cycle; the fundamental frequency of the AC component depends on the heart rate and is superimposed onto the DC component.
The interaction of light with biological tissue can be quite complex and may involve scattering, absorption and/or reflection. Anderson and Parrish examined the optical characteristics and penetration depth of light in human skin [5]; within the visible region, the dominant absorption peak corresponded to the blue region of the spectrum, followed by the green-yellow region (between 500 and 600 nm) corresponding to red blood cells. The shorter wavelengths of light are strongly absorbed by melanin. Water absorbs light in the ultraviolet and longer IR regime; however, red and near-IR light pass easily.
As a result, IR wavelengths have been used as a light source in PPG sensors. Blood absorbs more light than the surrounding tissue. Therefore, a reduction in the amount of blood is detected as an increase in the intensity of the detected light. The wavelength and distance between the light source and photodetector (PD) determine the penetration depth of the light. Green light is suitable for the measurement of superficial blood flow in skin. Light with wavelengths between 500 and 600 nm (the green-yellow region of the visible spectrum) exhibits the largest modulation depth with pulsatile blood absorption. IR or near-IR wavelengths are better for measurement of deep-tissue blood flow (e.g., blood flow in muscles).
Thus, IR light has been used in PPG devices for some time [6]. However, green-wavelength PPG devices are becoming increasingly popular due to the large intensity variations in modulation observed during the cardiac cycle for these wavelengths [7–9]. A green LED has much greater absorptivity for both oxyhaemoglobin and deoxyhaemoglobin compared to infrared light. Therefore, the change in reflected green light is greater than that in reflected infrared light when blood pulses through the skin, resulting in a better signal-to-noise ratio for the green light source.
Several green-light-based photoplethysmographs are available commercially. For example, MIO Global has developed the MIO Alpha in cooperation with Philips; this measures the electrocardiogram (ECG) with 99% accuracy, even while cycling at speeds of up to 24 kmph [10]. For daily use, Omron has developed a green light pulse rate monitor (HR-500U, OMRON, Muko, Japan). Furthermore, the use of video cameras using the signal based on the red green blue (RGB) colour space has been considered, as shown in Section 3.3. The green signal was found to provide the strongest plethysmographic signal among camera RGB signals [11,12]. Haemoglobin absorbs green light better than red and green light penetrates tissue to a deeper level than blue light. Therefore, the green signal contains the strongest plethysmographic signal.
The location of the LED and PD is an important design issue that affects the signal quality and robustness against motion artifacts. Therefore, suitable measurement sites must be located to optimize sensor performance. PPG sensors are commonly worn on the fingers due to the high signal amplitude that can be achieved in comparison with other sites [13]. However, this configuration is not well suited to pervasive sensing, as most daily activities involve the use of the fingers. In recent years, different measurement sites for PPG sensors have been explored extensively, including the ring finger [14], wrist [15,16], brachia [9,17,18], earlobe [19–21], external ear cartilage [22–24], and the superior auricular region [25–27]. In addition, the esophageal region has been used in clinical practice [28–30]. Commercial clinical PPG sensors commonly use the finger, earlobe and forehead [31]. In addition, use of a glass-type wireless PPG has been examined
The perfusion values of 52 anatomical sites in healthy subjects showed that the fingers, palm, face, and ears offer much higher perfusion values compared with other measurement sites [33]; the transmitted PPG signal amplitude from the earlobe provides the largest perfusion value. In addition, earlobe sensors are easy to fabricate, and have become popular as pulse rate monitors (Table 1). However, a spring-loaded ear-clip, although effective, can become painful over extended monitoring periods. There was little improvement in the wearable earlobe PPG sensor design until the development of micro-electromechanical system (MEMS) technology. MEMS facilitated the fabrication of a lightweight, comfortable, fully integrated, self-contained sensor earpiece. For example, an earring PPG sensor with magnetic attachment to the earlobe was developed that allowed good contact for monitoring during physical activity.
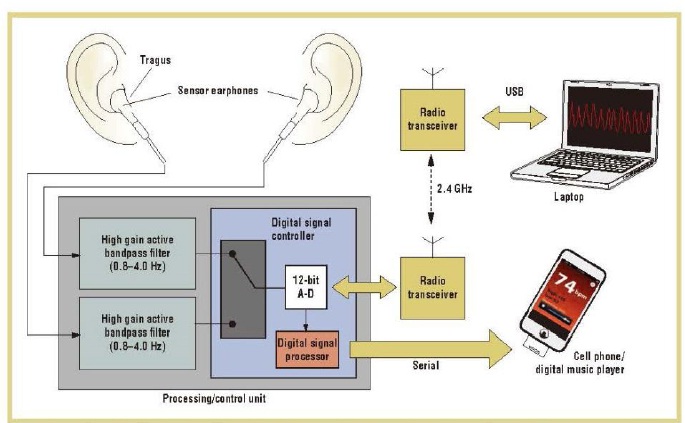
Earphone/earbud PPG sensors are also available and provide greater comfort for the user. In this design, a reflective photosensor is embedded into each earbud, as shown in Figure 3. The sensor earbuds are inserted into the ear and positioned against the inner side of the tragus to detect the amount of light reflected from the subcutaneous blood vessels in the region. The PPG sensor earbuds look and work like a regular pair of earphones, requiring no special training for use
A headset with an ear-clip, transmission-type PPG sensor allows continuous, real-time monitoring of heart rate while listening to music during daily activities. In addition, the proposed headset is equipped with a triaxial accelerometer, which enables the measurement of calorie consumption and step-counting. However, over the course of a variety of daily activities (e.g., walking, jogging, and sleeping), the PPG sensor signal may become contaminated with motion artifacts [20]. The most common commercially available PPG sensor is based on finger measurement sites.
Finger sites are easily accessed and provide good signal for PPG sensor probes. For example, a ring sensor can be attached to the base of the finger for monitoring beat-to-beat pulsations. Data from the ring sensor are sent to a computer via a radiofrequency transmitter, as shown in Figure 4. To minimize motion artifacts, a double ring design was developed to reduce the influence of external forces, acceleration and ambient light, and to hold the sensor gently and securely to the skin, so that the blood circulation in the finger remained unobstructed.
Experiments have verified the resistance of the ring sensor to interfering forces and acceleration acting on the ring body. Benchmark testing with FDA-approved PPG and ECG sensors revealed that the ring sensor is comparable in the detection of beat-to-beat pulsations, despite disturbances
Wristwatch-type pulse oximetry and blood pressure sensors have been developed and commercialized by several companies. These devices, although much easier to wear, are not usually used in clinical settings, due to several technical issues. However, a novel PPG array sensor module with a wristwatch-type design has been developed. The proposed module measures the PPG signal from the radial artery and the ulnar artery of the wrist, whereas previous methods obtained signals from the capillaries in the skin.
Phototransistors and IR-emitting diodes were placed in an array format to improve the PPG signal sensitivity and level of accuracy. Various arrays were considered for optimization. A conductive fiber wristband was used to reduce external noise. In the experiments, the proposed module was assessed and compared with the commercially available product produced by BIOPAC [16]. A reflective brachial PPG sensor has also been examined. Although the pulse amplitude is lower than those from the finger and earlobe, the PPG pulse waveforms from regions in the vicinity of a human artery could be detected and measured easily [17].
Forehead sensors have shown greater sensitivity to pulsatile signal changes under low perfusion conditions, compared with other peripheral body locations [31]. The thin-skin layer of the forehead, coupled with a prominent bone structure, helps to direct light back to the PD. Sensor placement on the forehead has been shown to result in decreased motion artifacts during certain types of physical activity.
Glass-type PPG sensors have also been investigated. A reflectance mode-PPG sensor, equipped with an accelerometer for detecting kinetic signals and a wireless controller for transmitting both signals to the receiver installed on the frame of the glass, was used to obtain PPG data from a user performing alternating sitting and sit-to-stand motions—the correlation between the peak-to-peak intervals in the signals of a BIOPAC device and the developed device was 97.5% and 87% for sitting and the sit-to-stand motion, respectively, given 100% data transmission without error
Photoplethysmography signals with an averaged AC/DC ratio of 0.001–0.01 and 10% relative strength compared with the finger-based approach were recorded from superior and posterior auricular skin [25]. An automated motion cancellation algorithm was designated to remove motion artifacts. This algorithm first determines the step frequency using the induced current readings from the reception channels.
When the LEDs were switched off, the outputs of the PDs were dominated by the motion artifact. The step frequency band from the reception channel outputs was then removed when the LEDs were switched on—this was achieved using notch filtering within the step frequency span (0.2 Hz, or 6 BPM). The remaining spectrum peak represented the heart rate frequency peak. The integrity of the PPG signal and heart rate detection accuracy were then evaluated. The results indicated that using the proposed passive motion cancellation, the device was able to reliably detect heart rate during periods of both rest and moderate exercise
Wearable PPG sensors have become very popular. Although a great deal of progress has been made in the hardware and signal processing, an acceptable wearable PPG sensor device has yet to be developed. Green light sources in PPG sensors minimize motion artifacts. Several filters and algorithms have been examined to mimic daily activities on limited time scales. However, better accuracy and reproducibility of real environments are required to eliminate motion artifacts. Further research is needed for the development of practical wearable PPG pulse rate monitors and pulse oximeters.
1. Togawa, T.; Tamura, T.; Öberg, P.Å. Biomedical Sensors and Instruments, 2nd ed.; CRC Press: New York, NY, USA, 2011; pp. 19–190.
2. Challoner, A.V.J. Photoelectric plethysmography for estimating cutaneous blood flow. In Non-invasive Physiological Measurement; Rolfe, P., Ed.; Academic Press: Oxford, UK, 1979; Volume 1, pp. 127–151.
3. Kamal, A.A.R.; Harness, J.B.; Irving, G.; Mearns, A.J. Skin photoplethysmography—A review. Comput. Methods Programs Biomed. 1989, 28, 257–269.
| Are you interested in this topic.Then mail to us immediately to get the full report.
email :- contactv2@gmail.com |