





Published on Nov 30, 2023
The day-to-day human activities and industrial revolution have influenced the flow and storage of water and the quality of available fresh water. Dyes are used in textiles, food and beverage industries and printing processes. The total dye consumption of the textile industry worldwide is in excess of 107 kg per year and approximately 1 million kg/yr of dyes are discharged into water streams by the textile industry. Dye producers and users are interested in stability and brightness and, consequently, are producing dyestuffs that are more difficult to degrade after use. Unless properly treated, the dyes present in wastewaters can affect photosynthetic activity due to reduced light penetration and may also be toxic to certain forms of aquatic life. Water has different sources like ground water, surface water, etc. and therefore the pollutants are also different. Chemical, textiles, tannery industries etc. cause high rate of pollution. A number of techniques such as electrochemical coagulation, photo catalytic de-colorization,electrochemical oxidation, adsorption and microbiological decomposition have been proposed from time to time for the removal of different types of dyes from water sources in an economic fashion.
The electrochemical technique has found wide acceptance for water treatment, because it currently offers promising approaches for the prevention of pollution. Use of graphene based nanomaterial in electrochemical technique is explored in this project. Experiments have been conducted to find the efficacy of the technique for different concentration of pollutants. Agricultural pollutants and the pollutants present in open water sources have been studied and it is found that the method is equally adaptive to these requirements. Thus, the method suggested finds its application in water supply as well as irrigation systems.
Keywords : Graphene, Dyes, Nanomaterials, Water Treatment
Preparation of electrochemical sensors using nano size graphene.
Setting up of measurement method for effective modification of the electrode surface and studying its surface morphology using various techniques like SEM (Scanning Electronic Microscopy), XRD (X-Ray Diffraction) and AFM (Atomic Force Microscopy) etc.
Optimization of electrochemical parameters like supporting electrolyte pH, accumulation time, modifier amount, temperature effect, scan rate for the determination.
Study suitability of the application for water purification with different concentration of pollutants in the sample.
A solution of congo red are prepared by dissolving 0.174g powdered sample in 25 ml double distilled water .The effect of supporting electrolyte was studied by using 0.2M phosphate saline (PBS) solution of different pH ranging from 3.0-11.2.
Different types of buffers like Britton- Robinson, Phosphate, Acetate, Citrate buffers are prepared.
Known amounts of pollutants (soils containing pollutants) sample which are available naturally and in the market are dissolved in known volume of solvent.
Fabricated sensor is given a pretreatment which is used in the analytical solution prepared using water and the pollutants either in the form of adding known amount of agricultural soil (containing pollutants) or those available in the market.
Electrolysis tests are conducted on the samples so prepared and observation as to the area of electrode affected, scan rate variation and pH variation.
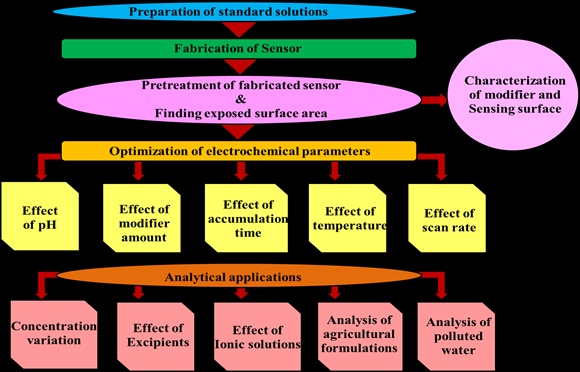
The area of the electrode was obtained by cyclic voltammetric method. The influence of pH of the analyte solution over the pH range 0.5-14 using buffer solution as a supporting electrolyte by CV at a preferred scan rate was determined.Useful information involving electrochemical mechanism is acquired from the relationship between peak current and scan rate. The effect of scan rate (υ) on the peak current and peak potential of analyteis evaluated. The transfer of electrons to or from the substrate is an activated process.
Based on the results obtained from experimental data i.e. pH variation, scan rate variation, area of the electrode, suitability of fabricating the graphene based electrodes for the treatment of water is determined. The method is found to be useful in identification and quantification of pollutants both for agricultural formulation and the dye pollutants in the water.Based on the result the method it is found that the method has a huge potential for the use in water treatment.
The experimental work carried out should become a base for the large scale application of the novel method of use of nano-materials for the treatment of water in flowing sources like rivers or stored in reservoirs. There is huge scope for this work as newer and newer materials in dying and tanning industries are invented creating newer pollutants for water.