





Updated on May 29, 2026
Spheroidal Graphite (S.G) Iron is the name given to that cast –iron, where the graphite is present in the matrix in the form of spheroid or globules or nodules. Hence it is also called as Nodular Iron and because of its exceptionally high ductility (with elongation some times as high as 22%) is also referred to as ductile iron.
The rapid growth of S.G iron and its high consumption are testimonials to its outstanding mechanical properties. Heat treatments such as austempering have further enhanced its properties. S.G iron is hence finding extensive application in number of the fields. After having basically replaced a number of gray iron and malleable iron casting they have made penetrating in roads, the forging industry such as crank shafts and cam shafts originally made by the forging technology are now being cast in iron. S.G iron is produced by the treatment of gray-iron with either cerium or magnesium. The more commercial of the two is magnesium and is now almost universally used for production of S.G iron. Use of cerium is restricted as it is a carbide forming element beyond 0.01%.
The first announcement of successful production of Spheroidal Graphite structures in grey cast irons was made by H.Morragh of British cast iron Research Association in the year 1948 at the American Foundrymen”s society’s Annual convention in Phladelphia, U.S.A.The process involved a treatment of low sulphur hyper- eutectic cast iron with cerium, which was added as mish metal shortly before casting. In same convention, T.M Wickenden of the International Nickel Company, U.S.A disclosed that their researchers had also found out a method of production of Spheroidal Graphite structure in the as cast condition by the introduction in the iron, either hyper- eutectic, of a small but effective amount of magnesium or a magnesium containing addition such as a nickel-magnesium alloy. Since then the latter process has been found to be a commercially viable process and is now universally used for the production of Spheroidal cast-iron.
The mechanism of graphite nodule formation however is still a matter of great controversy. Various theories have been put forward by various researchers but none of them have gained universal acceptance.
Basically S.G iron is produced by noduliging grey iron. Thus a melt of grey-iron is first of all desulphurised. Desulphurization has to be invariably carried out and recarburization has to be done if necessary. Once the base iron of required composition is ready, it is suitably treated for spheroidization. Post-inoculations are then carried out if required and the melt is poured in to the moulds as early as possible to avoid fading effect.
Engineering application of cast iron have been traditionally based upon gray (flake graphite) irons providing a range of tensile strengths between about 150N/mm2 and 400N/mm2 with recommended design stress in tensile application.

In contrast ductile irons have tensile strengths ranging from 350 to 1500N/mm2 with good elongation and high toughness. They now account for about 25% of iron casting production serving in safety critical applications where they have replaced steel casting, forging.
In short we can say that the highest mechanical properties of grey iron are the starting points for S.G iron castings.
The S.G. family iron has several interesting properties. These are given below
High strength (in some cases even greater than steel)
Adequate ductility.
Superior castability.
Excellent machinability.
Lower density then that of steel.
Superior surface lubrication properties.
Better damping characteristics.
The choice of chemical composition in alloys started with the aim of obtaining a carbon-free as-cast structure. The other factors that are considered are the effect of the various elements on the shape and distribution of graphite and the structure of the matrix. All factors are further affected by cooling rate.
It strongly promoted carbide free as cast structure. The carbon content for the commercial ductile iron is form 3.0 to 4.0%, although much narrower limits are usually desired. Nodule counts are directly affected by the carbon content greater number of spheroids formation at the higher carbon contents. Increasing the carbon content also increases castability by improving fluidity and feeding. The carbon –content level should be co-related with the carbon equivalent formula:-
CE = %C + 1/3 %Si + 1/3 %P
Carbon equivalents greatly is excess 4.3 promote the development and growth of graphite spheroids. Since graphite is far less dense than molten iron these spheroids may be become buoyant and float toward the cope surface of casting, resulting in gross carbon segregation, floating, as this phenomenon is called is prevent in analyses having carbon equivalents greater than 4.60 and in section sizes greater than 1inch.
It is a very strong promoter of carbide free as-cast structure. Apart from promoting graphite and refining its distribution, silicon is the most potent beneficial element in increasing strength and up to 4% will also increase as-cast ductility. Silicon being ferrite stabilizer, increases hardness, particularly in the annealed condition. Silicon also influences the distribution of graphite spheroids. The higher the silicon content, the higher the nodule count and the more the ferrite content. However, higher silicon content is known to promote chunky type graphite resulting in deterioration in properties of heavy ductile iron castings other potentially objectionable influences of increasing silicon content are reduced impact energy, increasing impact transition temperature and decreased thermal conductivity. The general range for commercial production is specified at around 1.8% to 2.8%.
The only single aim in selection of the manganese percentage is to avoid as-cast carbide. It must be preferably kept below 0.5%. A side benefit of lowered manganese content is the lowering of the tendency of hydrogen pick- up and there by minimization of the danger of pin hole. It must be noted that the manganese content must never be chosen with the aim of controlling the matrix structure.
The control of sulphur for the production of the S.G iron is highly essential. If base metal contains higher than 0.015% S are used in production of S.G iron, uneconomical amounts of magnesium or other nodulizing agents would be required and problems such as dross defect control get magnified. The base iron in-mould process should have sulphur contents within the 0.01% range.
Phosphorus decreases ductility, toughness and weldability and it is detrimental to casting soundness. For these reasons most specifications tolerate a maximum of 0.03% only. It is however, a good practice to keep phosphorus content below 0.04%. In certain cases where reduced creep rate, increased thermal conductivity and abrasion resistance are required, the phosphorus content should be much more.
Magnesium is added for nodularization generally in the alloyed form. It has been extensively reported in literature that the residual magnesium content in the S.G iron formed should be between 0.02% to 0.06% while calculating the amount of magnesium alloyed, required factors such as temperature of treatment, the efficiency of the treatment process and the percentage of the magnesium in the alloy will have to be taken in to consideration.
The aim when production good quality ductile iron is to produce an iron having a fine distribution of well formed nodules in the structure. The process of nodular graphite may reduce mechanical properties, the reduction depending of non–nodular or flake graphite which may present ISO 945 and ASTM 247 standard classify graphite according to a schematic presentation of the observed structure and the ASTM standard contains enable control personnel to assess graphite nodule number and size rapidly.
Small amount of element such as lead, bismuth, antimony and titanium singly or in combination prevent magnesium having the desired modularizing effect and hence are often referred to as subversive or interfering elements. The effects of subversive elements are cumulative. Small amounts of two or more subversive elements present in amounts which individually have no signification effect, together may adversely affected the formation of nodular graphite. The subversive effect of some elements:-
Particularly titanium, lead and bismuth increasing as the size of the cast section increases. For this reason acceptable levels can not be quoted and the effects of using furnace charge materials controlling subversive elements can be evaluated by determination of the effect on graphite structures in test bar or casting. When the elements are present individually, subversive effects are likely to occur if the following levels are acceded lead 0.0005%, bismuth 0.002%, antimony 0.004 and titanium 0.1%.
The effect of subversible elements can be neutralized by the adding of a small amount of cerium (0.002 to 0.005%) in addition to the magnesium.
Cerium contents above about 0.01% will results in a reduce nodule number and increasing risk of carbide formation, so it is important not to add too much.
The relationship of melting practice to the type and amount of spheroiding alloy used is of importance to casting and its physical properties
Cupola melting is the most common methods of melting for melting for ductile iron, moreover electrical induction furnace are in use in a number of foundries.
Using acid cupolas necessitates close control over charge materials and coke since the acid slag produced are not capable of reducing the sulpher content of the iron. This result in sulpher contents of 0.06 to 0.12 present, which, if not lowered, necessitates, the use of increasing amounts of spheroidizing alloy. Acid cupola melting, however, is capable of controlling the readily oxidizable elements in the charge, such as chromium and manganese. Since it is a more oxidizable process than the basic cupola operation. Because of the moderate carbon pick up in acid base cupola melting and the desired base iron chemistry , the use of pig iron of return is some what limited close composition control and high metal temperatures, however can be produce without the need of a hot blast.
Basic cupola melting is characterized by the definite advantage of sulpher control. Average sulpher content of the basic melt before spheradizing ranges from 0.025 to 0.035%. this decreased sulpher level in the melt is obtained at the expense of higher operating cost , higher silicon losses during melting , less effective temperature and composition control and a greater carbon pickup during melting.
Electrical melting has provided to be a clean and reliable melt. It has also offered the greatest flexibility for melting irons of different grades.
Electrical arc furnaces have been found to be less popular than induction heated ones. Their batch type operation and noise pollution have been the contributing factors. The facts that it has found some application both as a primary melter and as a duplexer is mainly due to its time prove reliability.
Electrical induction furnace have been the most common melting units for the production of S.G iron , both in simplex operation in small foundries and as duplexing units. The coreless type is being used for primary melting and a channel furnace is being used for duplexing .High frequency units have been found to be good form the metallurgical point of view but are associated with high costs. However, where furnace capacities under one tone are desirable and speed of melting is a consideration, than the high frequency is to be preferable. The coreless line frequency induction furnace has been found to be the best possible furnace for melting of base iron for S.G. iron production. The low frequency ensures better stirring thus resulting in homogenization melting
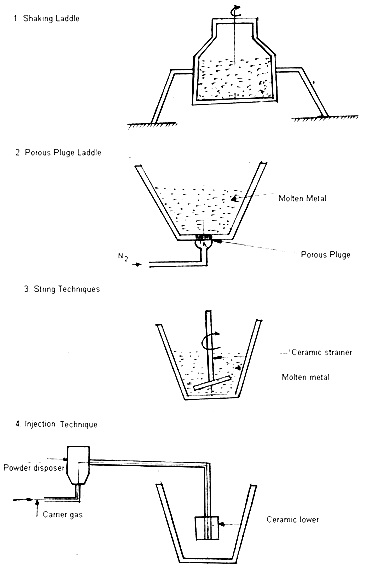
The application of the S.G iron have increased tremendously in recent times as can be seen from the list of the components which are being made.
Engine crank shaft
Brake caliper, disc brake anchor, brake anchor plate.
Machine- tool bed
Electrical insulator post and cap.
Steering Knuckle
Rack and pinion of steering assembly
Piston for impact drills.
Rolling mill rolls.
Moulding boxes and mould box clamps
Brake shoe for heavy duty brakes.
Glass moulds.
Spacer cage for rolling bearing.
Piston rings.
Wind mill items.
The ductile iron finds wide engineering application because of its excellent combination of castability and mechanical property. It has also very good machaniability, corrosion resistance, wear resistance and thermal shock resistance. It finds application in gears, dies, valve and pump bodies, pinion, crank shaft etc. It is a widely acceptable cast iron because of high strength and good ductility. The problems faced due to lower melting and boiling point of Mg have been encounter by using various magnesium treatment practices
1. Source book on “Ductile Iron”, ASM Publication, June 1977.
2. AFS Production of S.G. Iron Practice, 1976.
3. Fosico-ferrous Foundrymen, Handbook by R. Brown, 2000.
4. Principles of Metal Casting by Philip C. Rosenthal, 2003
5. ASM Hand Book, vol.15 (Casting), Ninth Edition, September, 1988.
6. Ductile Iron Production practice by BRCA, 1995.
7. Foundry Technology – Paul J. Mikelonis ASM and AFS, 1985.
8. Typical Microstructures of cast metal, by G. Lambert, 1966
| Are you interested in this topic.Then visit the below page to get the full report |