





Published on Apr 02, 2024
In this experiment you will measure the amount of stomach acid consumed (or neutralized) by various antacid tablets. If you have a favorite one, bring a package to the lab (one color only).
This experiment involves several steps. First the tablet is dissolved and an excess of acid of known concentration is added to the tablet. The solution is then briefly heated to insure that all of the antacid reacts. Finally, the remaining unreacted acid is titrated with base (NaOH) to determine the amount left over and hence, the amount that reacted with the tablet. The chemical reaction which occurs is:
NaOH + HCl -------> NaCl + H2O
A detailed description of the individual steps in the analysis follows.
1. Weigh an antacid tablet and transfer it to a 250 ml erlenmeyer flask. Record the weight of the tablets you are going to analyze.
2. Add 50.0 ml of hydrochloric acid solution (labeled 0.50 M) with a pipette to the flask containing the tablet.
3. If the tablet does not dissolve readily, cover the flask with a watch glass and boil gently for 5-10 minutes on a hotplate. Set it aside to cool. While it's cooling, weigh out another tablet of the same brand and proceed through steps 1 and 2
1. Check out a buret from the Stockroom. Test it to see if it is clean by filling with distilled water and letting it drain. If the water does not drain from the walls, wash carefully ($$$) with warm, soapy water using a buret brush. Rinse several times with distilled water.
2. Using a funnel fill the buret with base (sodium hydroxide) solution (labeled 0.5 M) to just above the top line.
3. Allow it to drain to just below the top line. If air bubbles remain in the tip, drain more out. If they still are in the tip, your instructor--who is lurking nearby, will be happy to get them out for you.
4. Add several drops of the indicator, thymol blue, to the cooled solution of antacid tablet containing unreacted acid. The solution will be red.
5. Read the initial volume of the buret using the graduations on the buret and bottom of the meniscus. It is often easiest to put a white piece of paper behind the buret to sight against. Record this as the initial volume for the first trial in your notebook in the results section.
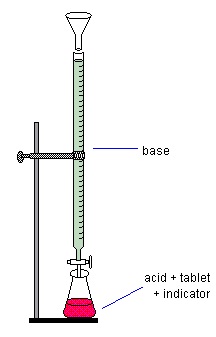
6. Add a small amount (less than one milliliter) of the sodium hydroxide to the flask. A spot of colorless solution may appear where the drops hit. With swirling, this area will disappear. The idea is to add the exact amount of the base solution needed to cause the solution in the beaker to change from red to very pale yellow and stay yellow for 15 seconds or more. This is harder than it may sound. The color change should be observed with the addition of only one or two drops of the base solution and you can't go back if you add too much. Watch the solution in the flask. As it gets more difficult to get rid of the pinkish color when you swirl the flask, add smaller amounts. Ideally, you should be adding it dropwise when you reach the point where color finally changes from red to yellow (endpoint). Patience! The first time is the hardest. Once you know how much it takes for a tablet, you can add slightly less in the next trial and add the last milliliter or so dropwise and you won't go past the endpoint. It may be necessary to refill the buret once.
7. Read and record the final volume. Calculate the total volume you used by subtracting the initial from the final volume. Record this, too, in your notebook.
8. As time permits, do one or two more trials. They should, as you practice, agree to within about 0.20 ml.
The HCl and NaOH used in this experiment are weak. They can be put into the sink with the water running. The finished titrations are not hazardous and can also be rinsed down the drain.
The number of grams of stomach acid that is neutralized can be calculated using the expression:
grams stomach acid per tablet = 10[ 25.0 - (ml base)(.50) ]
If, for example, it took 8.0 ml of base to titrate the sample, then
grams stomach acid = 10[ (25.0) - 8.0 (.50) ] = 210 g
Record the value for each of your samples and the average value with your results.
Does your tablet neutralize 47 times its weight in stomach acid? You can answer this question for your conclusion by dividing the mass of stomach acid by the mass of the individual tablet.
1. http://www.teachersfirst.com/lessons/forensics/ink-lab.html
2. www.reachoutmichigan.org/funexperiments/quick/csustan/mrsketch.htm