





Published on Apr 02, 2024
Aim is to Extract Nicotine Sulphate from Samples of Cigarettes
Tobacco is an agricultural product processed from the leaves of plants in the genus Nicotiana. It can be consumed, used as a pesticide and, in the form of nicotine tartrate, used in some medicines. It is most commonly used as a drug, and is a valuable cash crop for countries such as Cuba,India, China, and the United States. Tobacco is a name for any plant of the genus Nicotiana of the Solanaceae family (nightshade family) and for the product manufactured from the leaf and used in cigars and cigarettes, snuff, and pipe and chewing tobacco. Tobacco plants are also used in plant bioengineering, and some of the 60 species are grown as ornamentals.
The chief commercial species, N. tabacum, is believed native to tropical America, like most nicotiana plants, but has been so long cultivated that it is no longer known in the wild. N.Rrustica, a mild-flavored, fast-burning species, was the tobacco originally raised in Virginia, but it is now grown chiefly in Turkey, India, and Russia. The alkaloid nicotine is the most characteristic constituent of tobacco and is responsible for its addictive nature. The harmful effects of tobacco derive from the thousands of different compounds generated in the smoke, including polycyclic aromatic hydrocarbons (such as benzpyrene), formaldehyde, cadmium, nickel, arsenic, radioactive polonium-210, tobacco-specific nitrosamines (TSNAs), phenols, and many others.
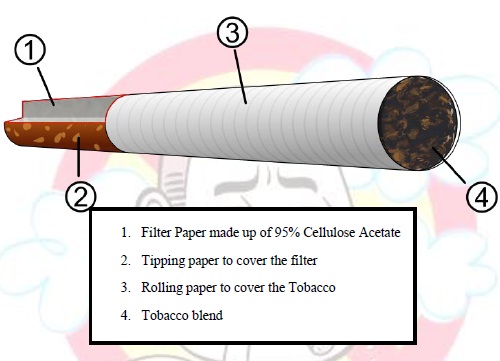
Tobacco is cultivated similarly to other agricultural products. Seeds are sown in cold frames or hotbeds to prevent attacks from insects, and then transplanted into the fields. Tobacco is an annual crop, which is usually harvested mechanically or by hand. After harvest, tobacco is stored for curing, which allows for the slow oxidation and degradation of carotenoids. This allows for the agricultural product to take on properties that are usually attributed to the "smoothness" of the smoke. Following this, tobacco is packed into its various forms of consumption, which include smoking, chewing, snuffing, and so on.
Most cigarettes incorporate flue-cured tobacco, which produces a milder, more inhalable smoke. Use of low-pH, inhalable, flue-cured tobacco is one of the principal reasons smoking causes lung cancer and other diseases association with smoke inhalation.
Nicotine is an alkaloid found in the nightshade family of plants (Solanaceae) that acts as a nicotinic acetylcholine receptor agonist. The biosynthesistakes place in the roots and accumulation occurs in the leaves of the Solanaceae. It constitutes approximately 0.6–3.0% of the dry weight of tobacco and is present in the range of 2–7 μg/kg of various edible plants. It functions as an antiherbivore chemical; therefore, nicotine was widely used as an insecticide in the past and nicotine analogs such as imidacloprid are currently widely used.
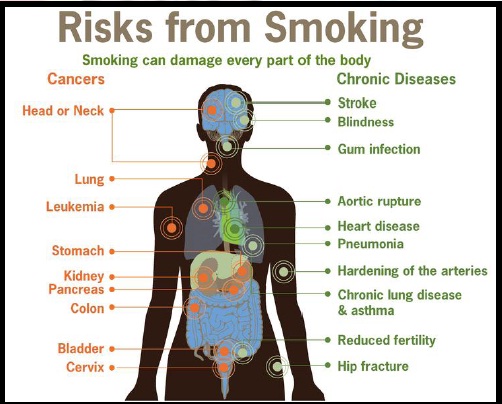
In low doses (an average cigarette yields about 1 mg of absorbed nicotine), the substance acts as a stimulant in mammals, while high amounts (30–60 mg) can be fatal. This stimulant effect is the main factor responsible for the dependence-forming properties of tobacco smoking. According to the American Heart Association, nicotine addiction has historically been one of the hardest addictions to break, while the pharmacological and behavioral characteristics that determine tobacco addiction are similar to those determining addiction to heroin and cocaine.
The nicotine content of popular American-brand cigarettes has slowly increased over the years, and one study found that there was an average increase of 1.6% per year between the years of 1998 and 2005. This was found for all major market categories of cigarettes.
250 ml Beaker
Filter Paper
Separating funnel
China Dish
Physical Balance
Glass Rod
Calcium Hydroxide [Ca(OH)2]
Kerosene as solvent
Concentrated Sulphuric Acid
Cigarettes
Collect different samples of cigarettes.
Remove the covers and collect the contents of the cigarettes on separate pieces of papers. Weigh 1 gram of each sample using a physical balance and label them. Place them in a dry place.
Dissolve each sample in Calcium Hydroxide solution taken in 100ml beakers. Stir them with the help of a glass rod.
Filter the different solutions after the Nicotine has fully dissolved. Collect them separately in separating funnels.
Add two test tubes of Kerosene Oil to each separating funner. Before pouring, ensure that the valve is closed.
After Pouring shake the mixture vigorously in order to mix the two liquids and leave it for two days without disturbing. Two layers – one heavy layer in the bottom and the second lighter layer in the top separates out.
Open the valve of the funnel and let the heavy layer out through the open valve. The lighter layer remains in the separating funnel.
Add half a test tube of concentrated Sulphuric Acid to each separating funnel. Again shake it vigorously in order to mix them and leave it for sometime.
Two layers are formed – one heavy (dark drown) layer at the bottom and another lighter (almost transparent) layer on the top.
Open the valve of the separating funnel and collect the heavier layer in a test tube.
Now transfer the contents into a china dish. Repeat the same procedure for the different samples and label them after the name of the cigarettes from which they have been obtained.
Heat the crystals in liquid form and ultimately subject them to cold water and leave them for a day or two.
Weigh the crystals hence obtained with the help of a physical balance.
The analysis showed relatively higher levels of nicotine in tobacco from beedis (0.17g) as compared to cigarettes (0.05g in Sample B and 0.06g in Sample A)

This study concludes that the nicotine content in tobacco contained in country cigarettes (beedi) is higher compared to the content in company manufactured branded cigarettes.
o www.en.wikipedia.com;
o http://emedicine.medscape.com/article/287555-overview;
o www.righthealth.com;
o www.netdoctor.co.uk;
o www.nicorette.com;
o “World Health Survey” by the World Health Organisation (WHO);
o “Practical Chemistry for Class XII” by Ratna Sagar Publications;
o “Science Reporter” July, 2010 edition;